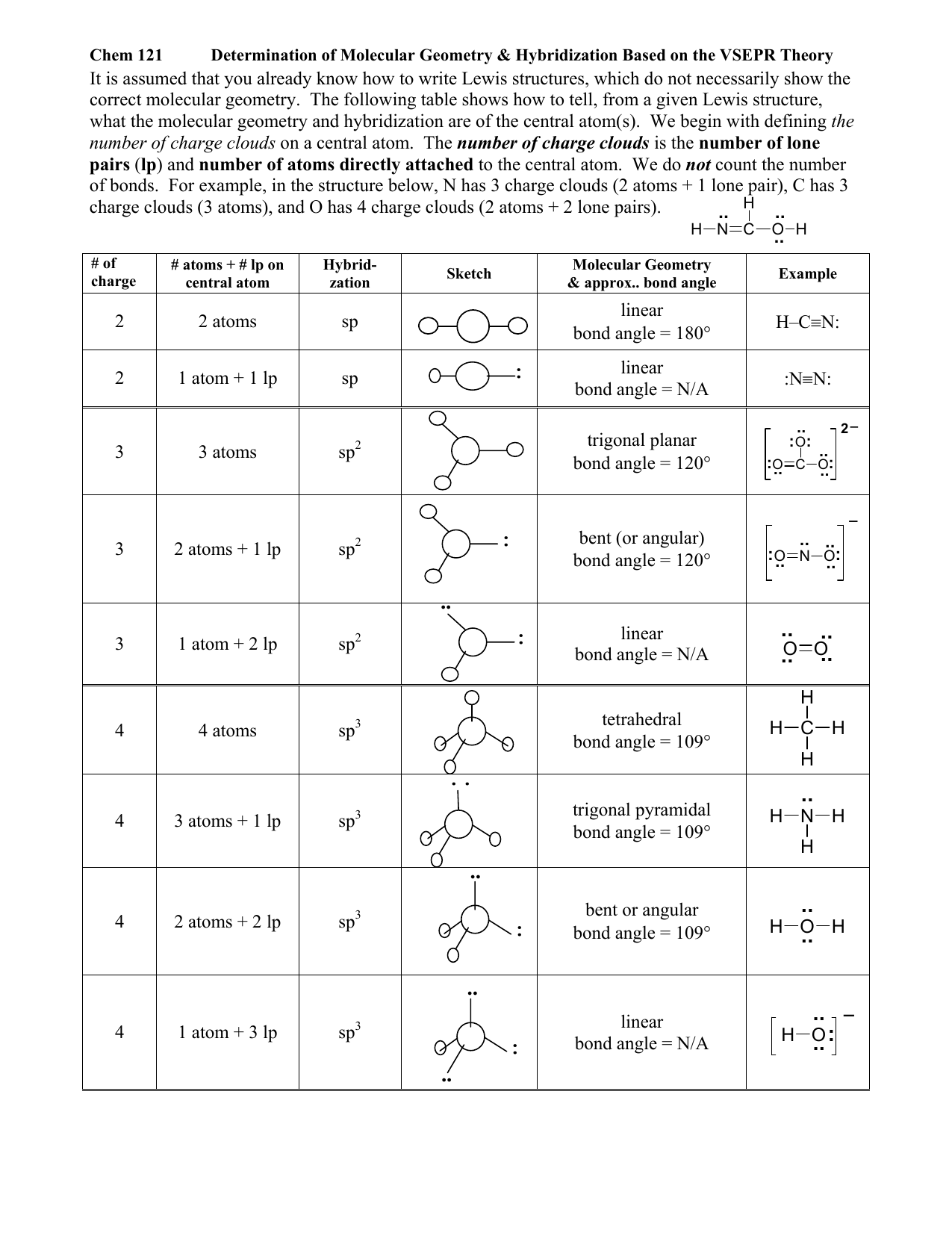
Molecular Geometry Chart With Hybridization
Molecular Geometry Chart With Hybridization - Web as we’ve seen, the ideal geometry for arranging four pairs of electrons is tetrahedral, which makes the hybridization of the central atom sp 3. Want to join the conversation? We will illustrate the use of this procedure with several examples, beginning with atoms with two electron groups. Web hybrid orbitals have very different shape from original atomic orbitals. Do you know that several factors make atoms come together and combine to form several different chemical compounds? The following table shows the equivalence: Predict the actual geometry of the molecule or ion ( 6 ). Sp 3 hybrid orbitals are oriented at bond angle of 109.5 o from each other. Describes the arrangement of atoms around the central atom with acknowledgment to only bonding electrons. Covalent bonds are formed by: In this video, we use both of these methods to determine the hybridizations of atoms in various organic molecules. Covalent bonds are formed by: The following table shows the equivalence: Sp 3 hybridization in methane. Web as we’ve seen, the ideal geometry for arranging four pairs of electrons is tetrahedral, which makes the hybridization of the central atom sp 3. Then, compare the model to real molecules! Read ratings & reviewsshop best sellersshop our huge selectionfast shipping In this compound valence electron will be as following: Predict the actual geometry of the molecule or ion ( 6 ). This type of hybridization is required whenever an atom is surrounded by two groups of electrons. This 109.5 o arrangement gives tetrahedral geometry (figure 4). Determine the hybridization of the central atom ( 7 ). Web we can find the hybridization of an atom in a molecule by either looking at the types of bonds surrounding the atom or by calculating its steric number. This type of hybridization is required whenever an atom is surrounded by. The steric number is how many atoms are bonded to a central atom of a molecule plus the number of lone electron pairs attached to that atom. Where six are arranged, around each oxygen atom in a way that one side has four valence electrons. Web hybrid orbitals have very different shape from original atomic orbitals. The angle between adjacent. Web hybridization directly correlates to molecular geometry! The lewis structure of h2s is as below. Web board index chem 14a molecular shape and structure determining molecular shape (vsepr) Web as we’ve seen, the ideal geometry for arranging four pairs of electrons is tetrahedral, which makes the hybridization of the central atom sp 3. So, before we start with organic chemistry,. The following table shows the equivalence: Determine the hybridization of the central atom ( 7 ). Web assign an ax m e n designation; How does molecule shape change with different numbers of bonds and electron pairs? Then, compare the model to real molecules! Web hybrid orbitals have very different shape from original atomic orbitals. Now to understand this we need to know the steps to draw a lewis structure at first. Web board index chem 14a molecular shape and structure determining molecular shape (vsepr) Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry cannot. Web lewis structure of o2. Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry cannot be explained using a vsepr approach. Web as we’ve seen, the ideal geometry for arranging four pairs of electrons is tetrahedral, which makes the hybridization of the central atom sp 3. Predict the actual geometry of. Lewis structures provide us with the number and types of bonds around a central atom, as well as any nb electron pairs. Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry cannot be explained using a vsepr approach. In this video, we use both of these methods to determine the hybridizations. Web lewis structure of o2. After calculating the electronic geometry from vespr we can determine the molecular geometry based on the bonding orbitals. Sp 3 hybridization in methane. Web as we’ve seen, the ideal geometry for arranging four pairs of electrons is tetrahedral, which makes the hybridization of the central atom sp 3. 2 molecular orbital (mo) theory. Web hybridization of an s orbital with all three p orbitals (p x , p y, and p z) results in four sp 3 hybrid orbitals. Now to understand this we need to know the steps to draw a lewis structure at first. Web hybridisation and geometry of molecules play a vital role in their reactivity. This type of hybridization. The steric number is how many atoms are bonded to a central atom of a molecule plus the number of lone electron pairs attached to that atom. Geometric · blankets & throws · microwaves · pyrex · teenage engineering This 109.5 o arrangement gives tetrahedral geometry (figure 4). Do you know that several factors make atoms come together and combine. If there are no lone pairs and all orbitals are bonding, then the molecular geometry is the electronic geometry. Web assign an ax m e n designation; This 109.5 o arrangement gives tetrahedral geometry (figure 4). Web predict the electronic geometry using all areas of electron density (or, effective electron pairs) and the ideal bond angles associated with this geometry ( 5 ). Web this vsepr chart shows you all of the common vsepr geometries, organized by the steric number and how many lone electron pairs they have. Usually, the electrons in the outermost shell of an atom, also known as valence electrons, take part in chemical. Now to understand this we need to know the steps to draw a lewis structure at first. Read ratings & reviewsshop best sellersshop our huge selectionfast shipping In this compound valence electron will be as following: Orbitals are combined in order to spread out electrons. Predict the actual geometry of the molecule or ion ( 6 ). Web hybridization increases the overlap of bonding orbitals and explains the molecular geometries of many species whose geometry cannot be explained using a vsepr approach. The steric number is how many atoms are bonded to a central atom of a molecule plus the number of lone electron pairs attached to that atom. This type of hybridization is required whenever an atom is surrounded by two groups of electrons. Then, compare the model to real molecules! 2 molecular orbital (mo) theory.VSEPR Chart With Hybridization
SO42 Lewis Structure, Molecular Geometry, Hybridization, and Polarity
Molecular Geometry And Hybridization Chart
MariePreAPChem Hybridization
Molecular Geometry Chart With Hybridization
No3 Lewis Structure Molecular Geometry And Hybridization
HYBRIDIZATION Teaching chemistry, Chemistry basics, Organic chemistry
C2H4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram
Xecl4 Lewis Structure Geometry Hybridization And Polarity guidetech
Hybridization and Hybrid Orbitals ChemTalk
Web Board Index Chem 14A Molecular Shape And Structure Determining Molecular Shape (Vsepr)
Web Hybridization Of An S Orbital With All Three P Orbitals (P X , P Y, And P Z) Results In Four Sp 3 Hybrid Orbitals.
Covalent Bonds Are Formed By:
Reactions Involve Making And Breaking Of Bonds!
Related Post:



2]%2B%2B4.%2BTetrahedral.%2Bsp3.%2B[Cd(NH3)4]2%2B.jpg)



