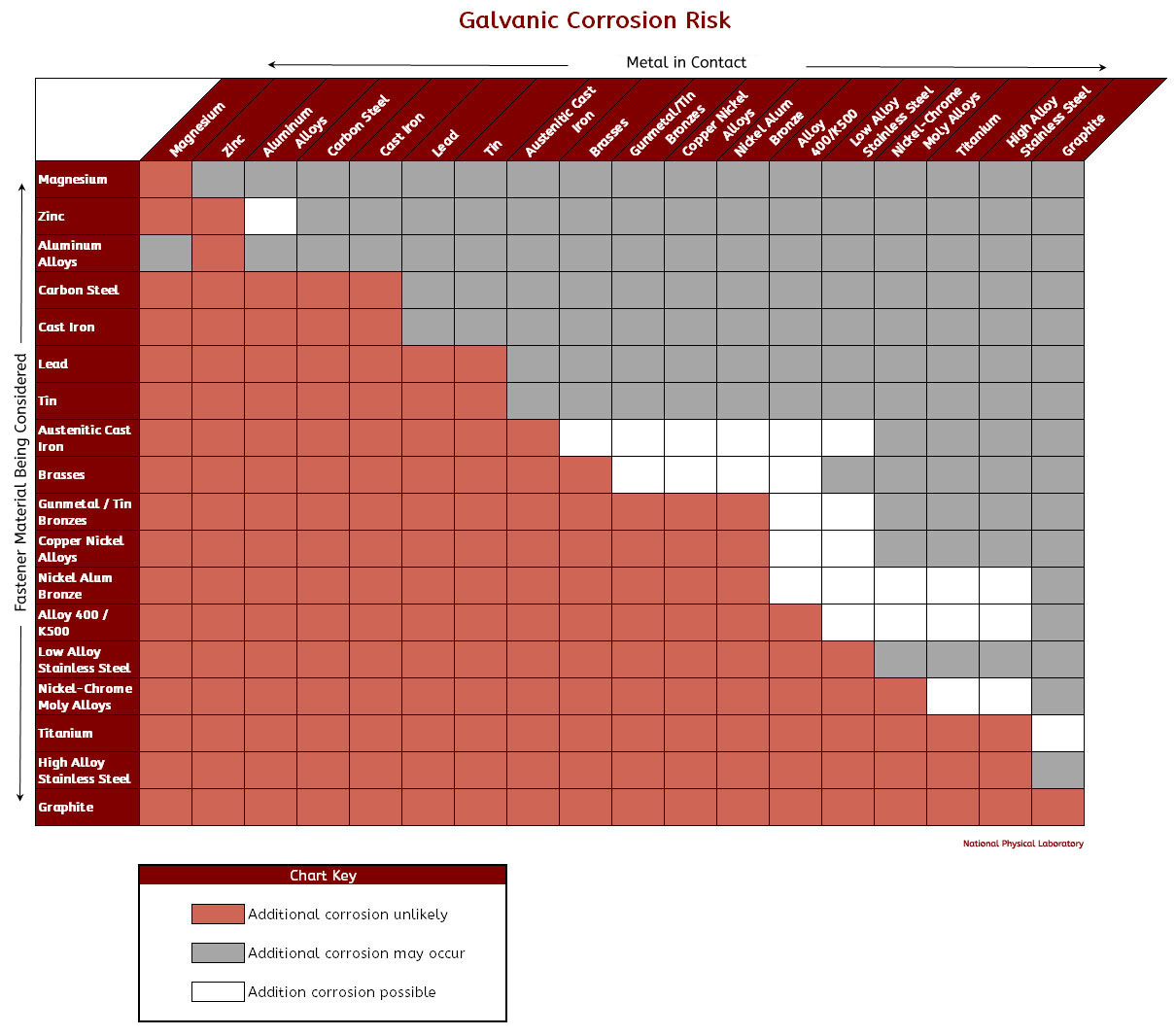
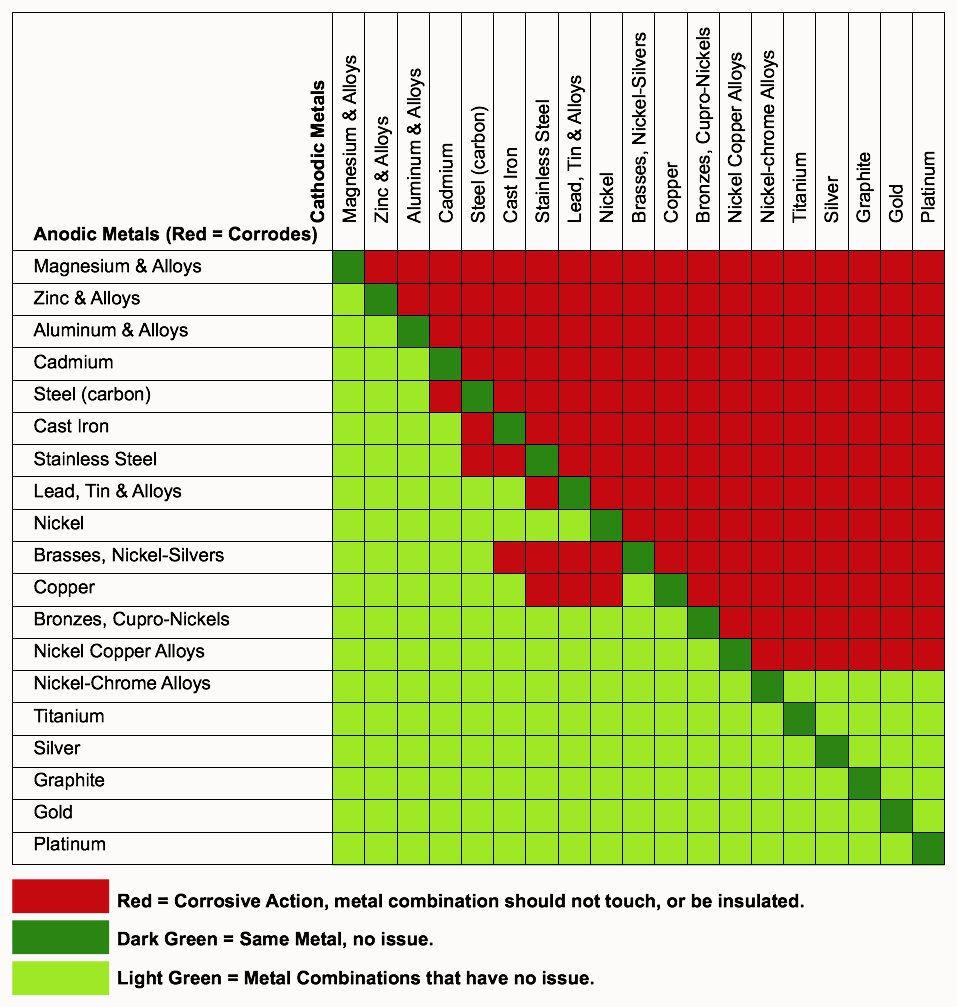
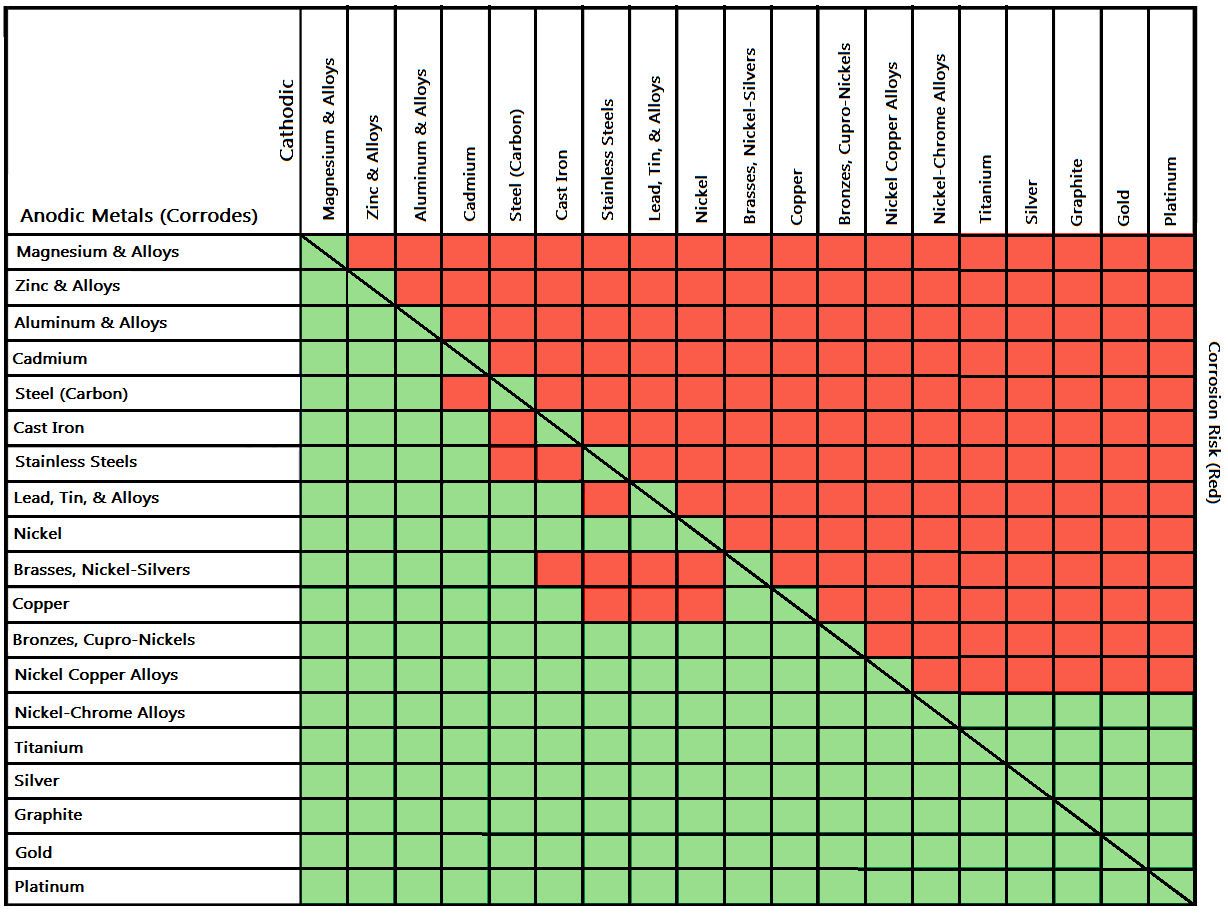
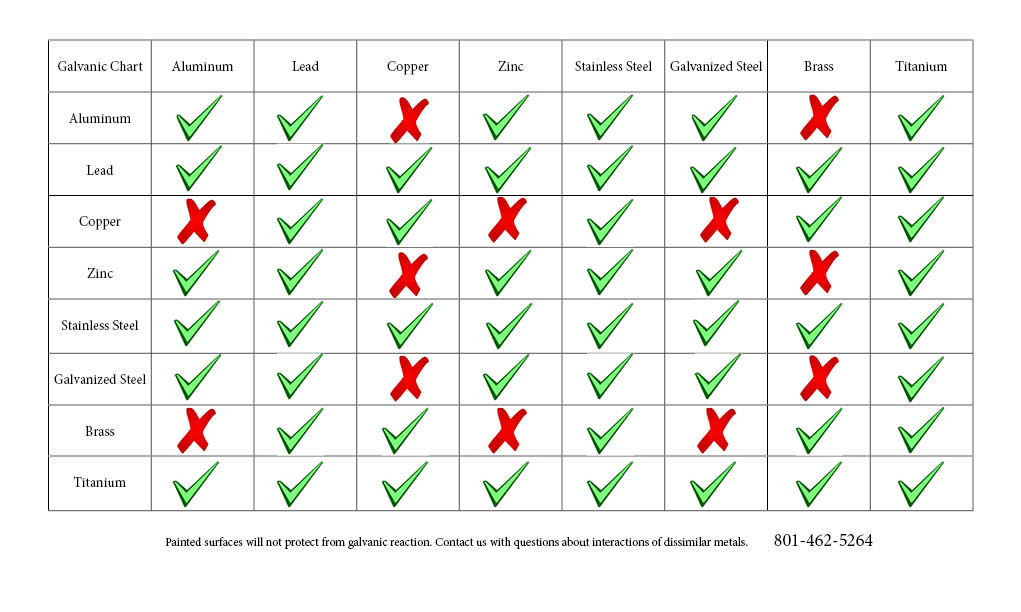
Galvanic Chart Corrosion
Galvanic Chart Corrosion - Web below is a galvanic reaction chart for dissimilar metals. This phenomenon is named after italian ph… This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Web the galvanic corrosion table ranks metals from the most “active” to the least active. Fastened of galvanic corrosion in joint, it’s. Web below is the galvanic corrosion chart, along with steps for using the chart properly to determine metal compatibility. Web the increased corrosion of the anode is called “galvanic corrosion.” galvanic corrosion is sometimes used to extend the life of materials (i.e. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Electric current flows from plus to minus. This can help you in the selection of the best. Electric current flows from plus to minus. Web galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the materials in contact with each other oxidizes or corrodes. This phenomenon is named after italian ph… This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal. Fastened of galvanic corrosion in joint, it’s. Zinc coatings on carbon steel and. Web the galvanic corrosion table ranks metals from the most “active” to the least active. The larger the separation distance in the electromotive chart between the two metals in contact, the. This can help you in the selection of the best. The most active metals in the galvanic corrosion chart, like aluminum, zinc, or. Web galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the materials in contact with each other oxidizes or corrodes. We consider two metals compatible if their emf. Zinc coatings on carbon steel and. Web below is a galvanic reaction chart for dissimilar. Web the increased corrosion of the anode is called “galvanic corrosion.” galvanic corrosion is sometimes used to extend the life of materials (i.e. Material selection is important because different metals come into contact with each other and. The below galvanic corrion chart or anodic index table shows anodic index for different materials. This chart is designed to assist in broadly. Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. However, understanding how it works and the steps that can be taken to protect against. Electric current flows from plus to minus. A similar galvanic reaction. Zinc coatings on carbon steel and. Web typically, the presence of an electrolyte (eg. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). Web when corrosion does occur, the anodic material is the most likely to corrode, whereas the cathodic material is the least likely to corrode. However, understanding how. The closer together the material are on the chart to the right, the less. Web galvanic corrosion is of particular concern in design and material selection. Water) is necessary to promote galvanic corrosion. Web by knowing the relationships of the metals in the series, galvanic compatibility can be determined, preventing the possible harmful effects of galvanic. Web the metal with. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. Material selection is important because different metals come into contact with each other and. Web the metal with the higher potential forms the anode and is corroded. A typical rule of thumb is that. We consider two metals compatible if their. Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. We consider two metals compatible if their emf. The most active metals in the galvanic corrosion chart, like aluminum, zinc, or. The below galvanic corrion chart. The closer together the material are on the chart to the right, the less. Web there are four elements necessary for corrosion to occur in a galvanic cell: This phenomenon is named after italian ph… This can help you in the selection of the best. Web the increased corrosion of the anode is called “galvanic corrosion.” galvanic corrosion is sometimes. Web the galvanic corrosion table ranks metals from the most “active” to the least active. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). This chart is designed to assist in. The most active metals in the galvanic corrosion chart, like aluminum, zinc, or. The closer together the material are on the chart to the right, the less. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). This phenomenon is named after italian ph… Electric current flows from plus to minus. Electric current flows from plus to minus. Material selection is important because different metals come into contact with each other and. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. We consider two metals compatible if their emf. This can help you in the selection of the best. Web galvanic corrosion is of particular concern in design and material selection. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). Web there are four elements necessary for corrosion to occur in a galvanic cell: Fastened of galvanic corrosion in joint, it’s. This phenomenon is named after italian ph… Web below is a galvanic reaction chart for dissimilar metals. The closer together the material are on the chart to the right, the less. Water) is necessary to promote galvanic corrosion. Web by knowing the relationships of the metals in the series, galvanic compatibility can be determined, preventing the possible harmful effects of galvanic. Zinc coatings on carbon steel and. Web the metal with the higher potential forms the anode and is corroded.Galvanic Corrosion [with Chart] EngineerExcel
Galvanic Corrosion SSINA
Galvanic Corrosion Chart PDF Corrosion Electrochemistry
Galvanic Corrosion Chart
Galvanic Action Corrosion Prevention Architect's Blog
Galvanic Corrosion Common Questions Answered
Galvanic Corrosion Chart Metals
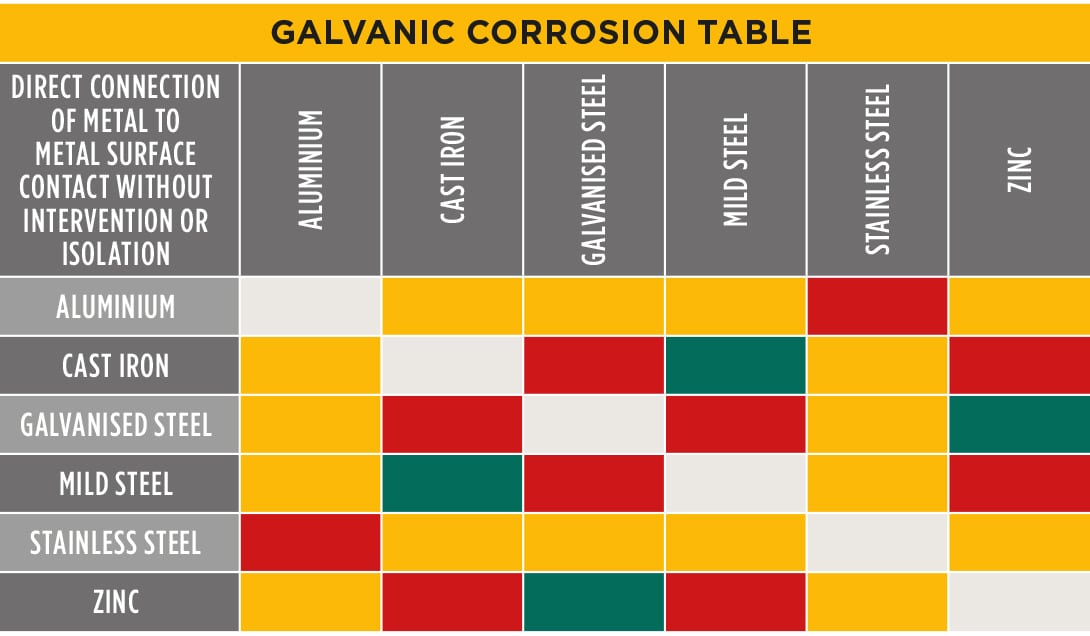
GALVANICCORROSIONTABLE CMP Products Limited
Galvanic Corrosion CMP Products Limited
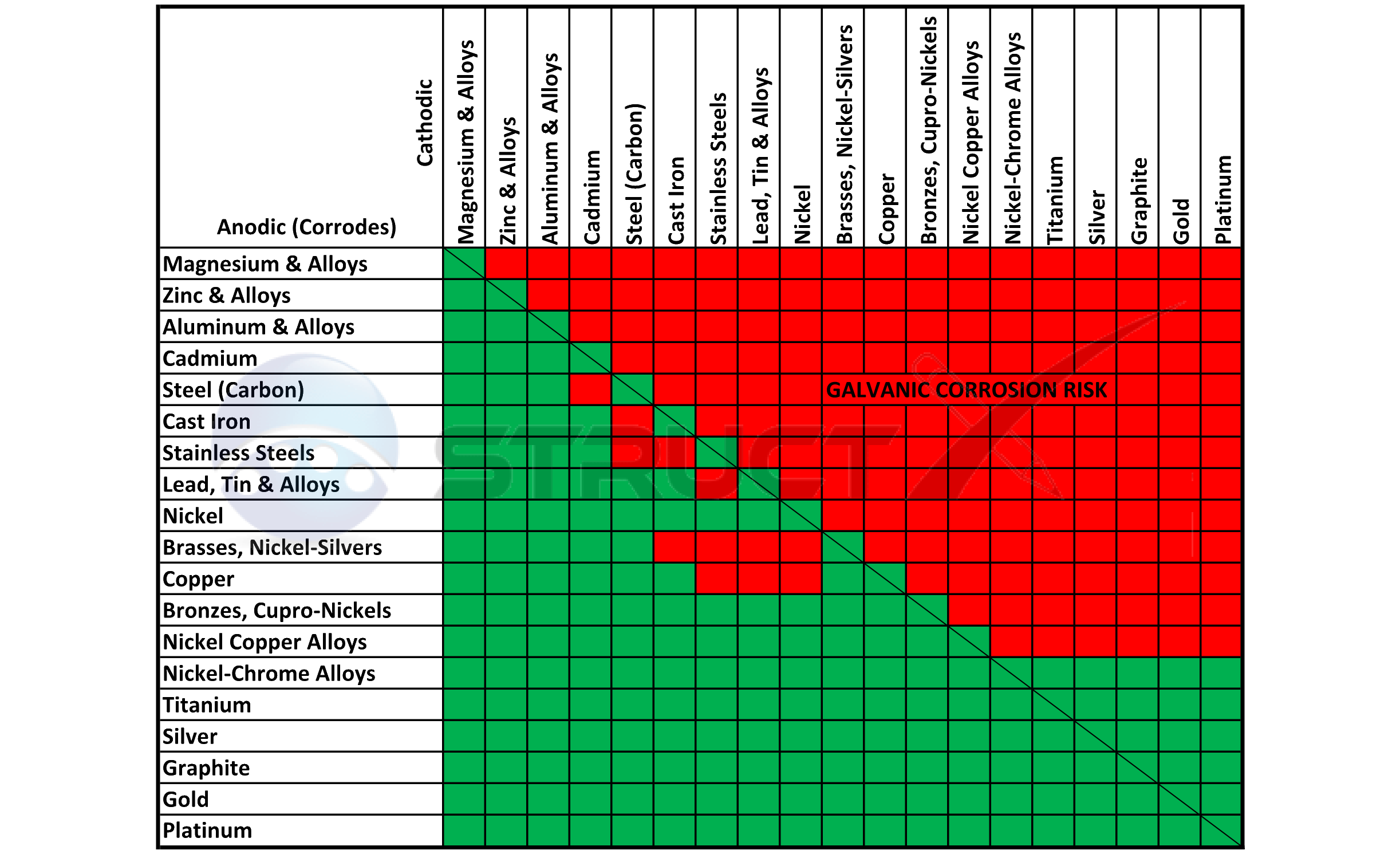
Galvanic Series (electrochemical series)
Web Below Is A Galvanic Reaction Chart For Dissimilar Metals.
Web To Minimize Galvanic Corrosion, Select Fasteners Based On Their Material Compatibility With The Substrates.
Web Below Is The Galvanic Corrosion Chart, Along With Steps For Using The Chart Properly To Determine Metal Compatibility.
Galvanic Corrosion (Also Called Bimetallic Corrosion Or Dissimilar Metal Corrosion) Is An Electrochemical Process In Which One Metal Corrodes Preferentially When It Is In Electrical Contact With Another, In The Presence Of An Electrolyte.
Related Post:
![Galvanic Corrosion [with Chart] EngineerExcel](https://engineerexcel.com/wp-content/uploads/2023/03/galvanic-corrosion-chart.png)