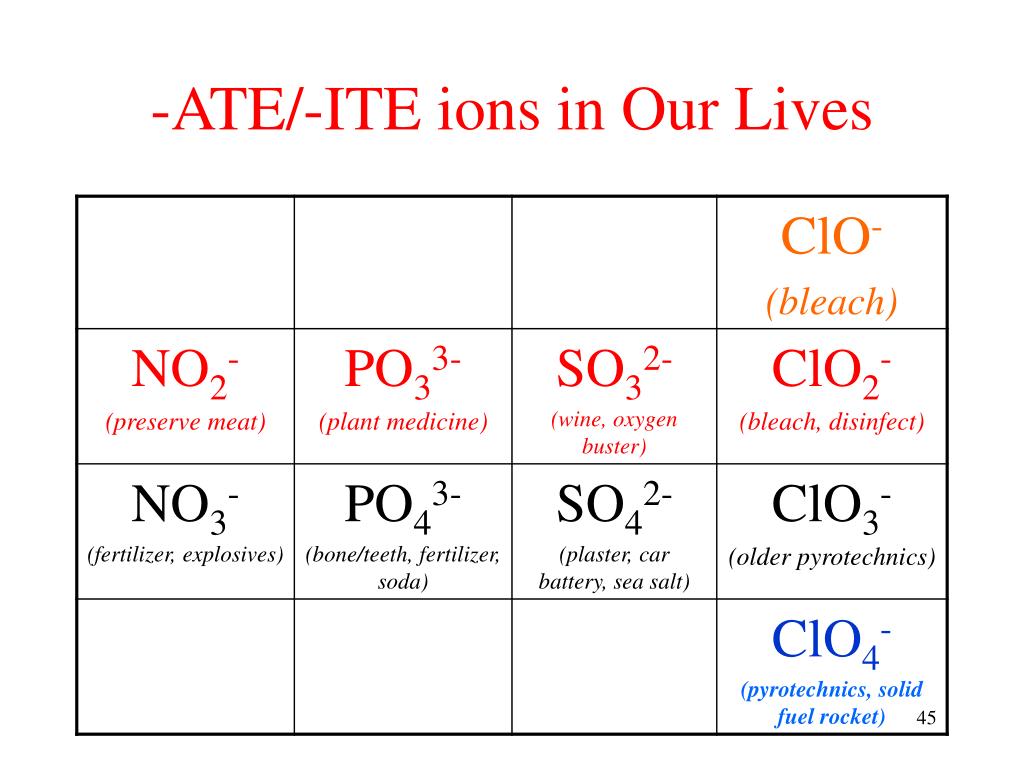
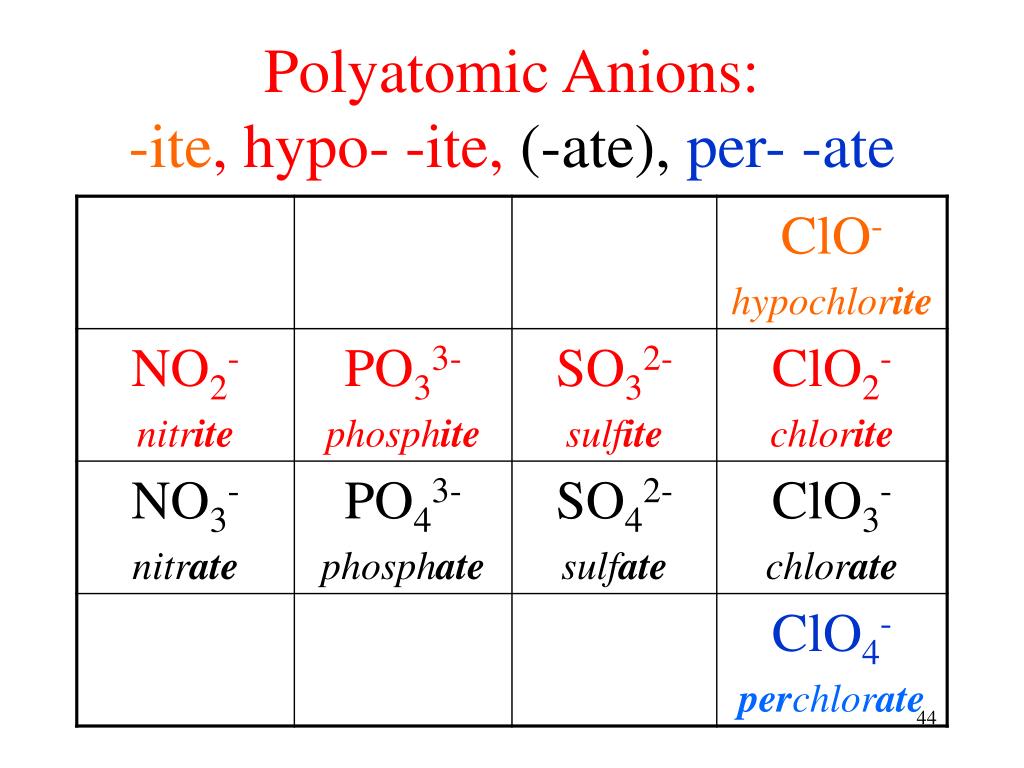
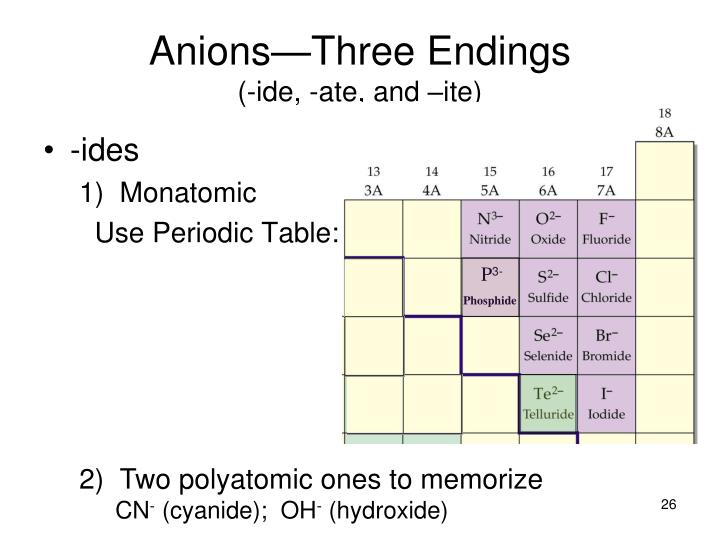
Ate Ite Ide Chart
Ate Ite Ide Chart - Let's say below is what you see written: It doesn't denote a specific number. For example, chlorine forms a chloride ion, so nacl is sodium chloride. These anions are called oxyanions. So no2 would be nitrite, and no3 would be. Web flow chart for naming simple inorganic compounds. It is the suffix of the. The flowchart is adapted from p. Some polyatomic anions contain oxygen. So, no2 is nitrite, and. The flowchart is adapted from p. Some polyatomic anions contain oxygen. Web so you see, there's a big difference between ending with ide or with ate. Web terms in this set (30) study with quizlet and memorize flashcards containing terms like ammonium, acetate, bromate and more. Learning a language means paying attention to patterns. So, no2 is nitrite, and. It is the suffix of the. When an element forms two. These anions are called oxyanions. Ending (suffix) is only for polyatomic anions that contain oxygen, when the polyatomic ion is contained in the following: These anions are called oxyanions. So, no2 is nitrite, and. When an element forms two. It is the suffix of the. Learning a language means paying attention to patterns. Web flow chart for naming simple inorganic compounds. Ending (suffix) is only for polyatomic anions that contain oxygen, when the polyatomic ion is contained in the following: Some polyatomic anions contain oxygen. It doesn't denote a specific number. For example, chlorine forms a chloride ion, so nacl is sodium chloride. These anions are called oxyanions. Web so you see, there's a big difference between ending with ide or with ate. Learning a language means paying attention to patterns. When an element forms two. The flowchart is adapted from p. Ending (suffix) is only for polyatomic anions that contain oxygen, when the polyatomic ion is contained in the following: Some polyatomic anions contain oxygen. When an element forms two. For example, chlorine forms a chloride ion, so nacl is sodium chloride. It is the suffix of the. The flowchart is adapted from p. For example, chlorine forms a chloride ion, so nacl is sodium chloride. Web terms in this set (30) study with quizlet and memorize flashcards containing terms like ammonium, acetate, bromate and more. It doesn't denote a specific number. Web flow chart for naming simple inorganic compounds. Learning a language means paying attention to patterns. So no2 would be nitrite, and no3 would be. Ending (suffix) is only for polyatomic anions that contain oxygen, when the polyatomic ion is contained in the following: When an element forms two. The flowchart is adapted from p. It doesn't denote a specific number. For example, chlorine forms a chloride ion, so nacl is sodium chloride. These anions are called oxyanions. Let's say below is what you see written: Learning a language means paying attention to patterns. For example, chlorine forms a chloride ion, so nacl is sodium chloride. These anions are called oxyanions. So, no2 is nitrite, and. Web terms in this set (30) study with quizlet and memorize flashcards containing terms like ammonium, acetate, bromate and more. When an element forms two. So, no2 is nitrite, and. Let's say below is what you see written: Web so you see, there's a big difference between ending with ide or with ate. For example, chlorine forms a chloride ion, so nacl is sodium chloride. Some polyatomic anions contain oxygen. It doesn't denote a specific number. For example, chlorine forms a chloride ion, so nacl is sodium chloride. It is the suffix of the. Learning a language means paying attention to patterns. Web terms in this set (30) study with quizlet and memorize flashcards containing terms like ammonium, acetate, bromate and more. Web so you see, there's a big difference between ending with ide or with ate. The flowchart is adapted from p. Let's say below is what you see written: When an element forms two. These anions are called oxyanions. So no2 would be nitrite, and no3 would be. Ending (suffix) is only for polyatomic anions that contain oxygen, when the polyatomic ion is contained in the following:Formula Making of Anions with suffix ide, ate, ite ( With Trick
PPT Molecules and Compounds Nomenclature PowerPoint Presentation
PPT Molecules and Compounds Nomenclature PowerPoint Presentation
PPT Breakdown of Topics PowerPoint Presentation ID6209332
PPT Chapter 6 “Chemical Names and Formulas” PowerPoint Presentation
ide ite ate YouTube
PPT Guide to Naming Ionic Compounds PowerPoint Presentation, free
HOW TO USE NOMENCLATURE YouTube
Ate Ite Ide Chart
Some Polyatomic Anions Contain Oxygen.
Web Flow Chart For Naming Simple Inorganic Compounds.
So, No2 Is Nitrite, And.
Related Post: